Professor Aneel Bhangu, Director, Surgical Data Institute, University of Birmingham, UK. a.a.bhangu@bham.ac.uk
DOI: 10.5281/zenodo.19237248
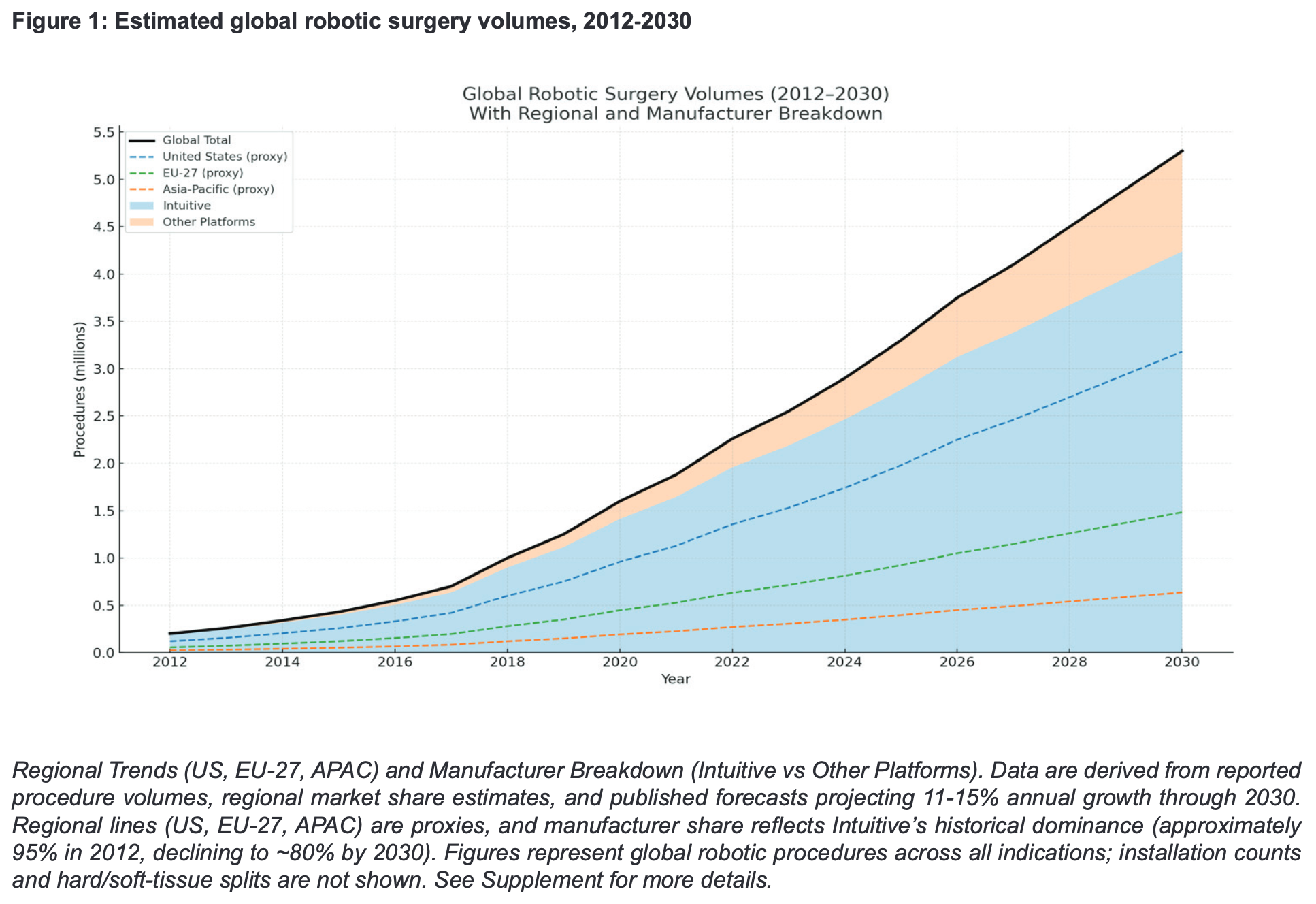
Robotic-assisted surgery has now progressed beyond early adoption, and across the world is moving from niche to mainstream. Best estimates from aggregated procedure counts and independent forecasts suggest compound annual growth of approximately 11–15% through to 2030, at least. The United States, the EU-27, and the Asia-Pacific region all show rising utilisation with similar trajectories. Manufacturer dynamics are also shifting, with Intuitive's market share easing from approximately 95% in 2012 to roughly 80% projected by 2030. In England, robot-supported procedures increased from fewer than 7,000 in 2014 to over 70,000 in 2023/24, and national targets now stand at around 500,000 operations per year by 2035. The policy question has shifted from whether to integrate robotics to how to scale safely, equitably, and cost-effectively, whilst generating decision-ready evidence. Early Value Assessment guidance from NICE has firmly linked conditional recommendations to structured evidence generation.
Soft-tissue robotics has been led for two decades by the da Vinci system, for which the field has much to be appreciative. Intuitive reported 2024 revenue of US$8.35 billion, a 17% year-on-year increase, with 84% derived from recurring sources, reflecting sustained procedure growth and consumable use. Many new entrants are now providing procedure-specific alternatives. CMR Surgical's modular Versius platform has received regulatory clearance in major markets and is in routine clinical use, with more than 30,000 procedures reported across over 30 countries. Johnson & Johnson's OTTAVA system has completed its first clinical cases within a formal trial, marking the beginning of human use. Medtronic's Hugo robotic-assisted surgery platform has expanded its European offering following CE mark approval for the LigaSure vessel sealing instrument, extending its role across gynaecological, general, and urological procedures. Alongside these systems, a growing number of soft-tissue robots from global manufacturers are entering development and early clinical use, marking a shift towards a genuinely competitive robotic surgery market.
In orthopaedics, diffusion has been equally substantial. Stryker's Mako platform has exceeded one million procedures across 35 countries, while Zimmer Biomet's ROSA and Globus Medical's ExcelsiusGPS continue to expand indications and installed base. Meta-analyses suggest improvements in radiographic alignment and outlier reduction after robotic knee arthroplasty, though consistent gains in functional outcomes or revision rates remain uncertain, identifying the need for long-term comparative data.
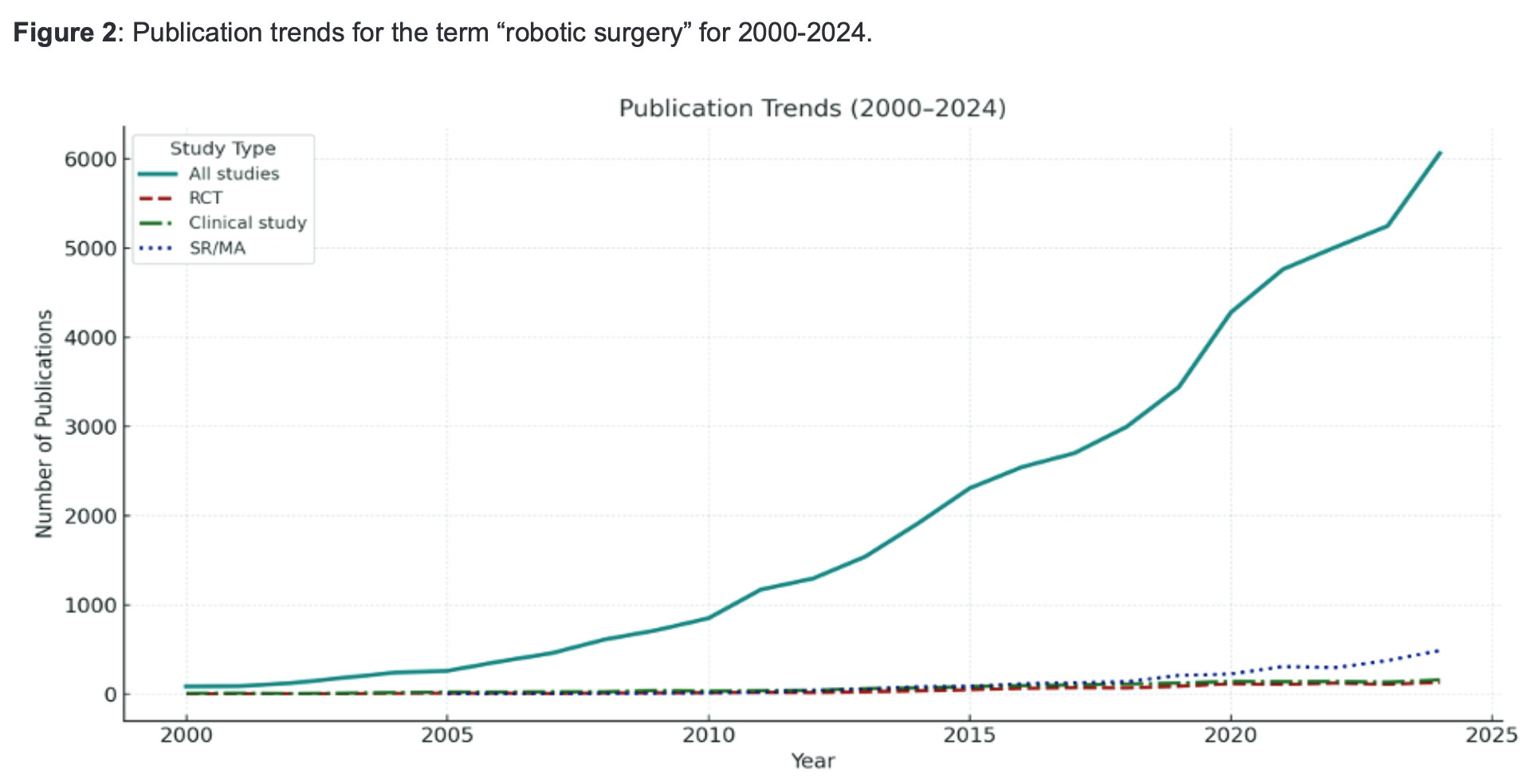
The literature base is expanding across specialties but remains dominated by case series and single-centre cohorts rather than multicentre randomised trials. Landmark trials, including ROLARR for rectal cancer and LACC for cervical cancer, illustrate the challenges of conducting pragmatic surgical trials in the context of learning curves and evolving systems. Orthopaedic trials and systematic reviews continue to mature but have not yet delivered uniform evidence of clinical superiority, despite gains in precision.
The concept of robotic plurality, or multiplatform robotics, describes service models in which teams train and practise across multiple platforms within a health system, enabling vendor competition, reducing over-dependence on a single supplier, and supporting platform-agnostic curricula. Plurality can facilitate procurement efficiency and resilience, while allowing modular, lower-footprint systems to extend access where capital constraints prevent flagship installations. For plurality to deliver value, three conditions are necessary: interoperable data allowing comparative outcomes across platforms; competency-based training and credentialling that are portable between systems; and adaptive technology assessments responsive to iterative hardware and software upgrades.
Equity considerations are important to the future for both providers and governments. Billions of people lack access to safe surgical care, and without explicit attention to equity, robotics risks deepening disparities between and within countries. Public systems in low- and middle-income settings face substantial capital and maintenance barriers, and adoption has clustered in private urban hospitals. Pragmatic models — lower-cost robots, digital laparoscopic assistants, and adjuncts for open surgery — could support equitable technology access if specified for maintainability, local technical support, and total cost of ownership. International collaborations should prioritise platform-agnostic training, open data standards, and outcomes that matter to patients rather than throughput alone.
Investment in robotics is typically described alongside opportunity costs relative to alternative capital projects and service delivery, although these costs may be smaller than expected and may not exist at all if the benefits are fully realised. Industry and health systems should develop decision-grade evidence on outcomes, costs, and wider benefits. NICE's conditional approvals provide a lever to align reimbursement with real-world evidence generation. More will be achieved if industry and research organisations work together to accelerate comparative effectiveness research.
The trajectory of robotic surgery is now sufficiently established that the field must move beyond adoption debates and address the harder questions: how evidence generation keeps pace with technology iteration, how training systems remain coherent across a multiplying platform landscape, and how the benefits of robotic surgery reach health systems and patient populations currently excluded from them. These are not peripheral concerns. They are the conditions under which robotic surgery consolidates its place as necessary infrastructure rather than remaining, in parts of the world, a novelty.
Previously published as: Bhangu A. The growth of robotic surgery: from novelty to necessary infrastructure. Impact Surgery. 2025;2(7):250–253. doi: 10.62463/surgery.293
Conflicts of interest: None declared.
References
- NHS England; Getting It Right First Time (GIRFT). Implementation of robotic-assisted surgery (RAS) in England. London: NHS England and GIRFT; 2025. Available from: https://gettingitrightfirsttime.co.uk/wp-content/uploads/2025/07/FINAL_NHS-England-and-GIRFT-implementation-of-robotically-assisted-surgery-in-England_17-07-2025.pdf
- NHS England. Millions to benefit from NHS robot drive. 11 Jun 2025. Available from: https://www.england.nhs.uk/2025/06/millions-to-benefit-from-nhs-robot-drive/
- Sollof J. NHSE projects 500k robotic-assisted operations a year by 2035. Digital Health. 11 Jun 2025. Available from: https://www.digitalhealth.net/2025/06/nhse-projects-500k-robotic-assisted-operations-a-year-by-2035/
- Royal College of Surgeons of England. Robotic-assisted surgery: a pathway to the future. London: RCS England. Available from: https://www.rcseng.ac.uk/standards-and-research/standards-and-guidance/good-practice-guides/robotic-assisted-surgery/
- National Institute for Health and Care Excellence. Robot-assisted surgery for orthopaedic procedures: Early Value Assessment (HTE22). London: NICE; 2025. Available from: https://www.nice.org.uk/guidance/hte22
- Intuitive Surgical, Inc. Annual report 2024. United States Securities and Exchange Commission filing. 2025. Available from: https://www.sec.gov/Archives/edgar/data/1035267/000103526725000099/a2024_intuitiveannualrepor.pdf
- CMR Surgical. CMR Surgical receives U.S. FDA Marketing Authorization for next-generation Versius Surgical System. 15 Oct 2024. Available from: https://www.globenewswire.com/news-release/2024/10/15/2963054/0/en/CMR-Surgical-receives-U-S-FDA-Marketing-Authorization-for-next-generation-Versius-Surgical-System.html
- Gapper J. British surgical robots should operate around the world. Financial Times. 2 Jun 2025. Available from: https://www.ft.com/stream/82458fd9-6374-45cc-b4f8-87afebeafbd1
- Johnson & Johnson MedTech. Johnson & Johnson MedTech announces completion of first cases with OTTAVA Robotic Surgical System. Press release. 14 Apr 2025. Available from: https://www.jnj.com/media-center/press-releases/johnson-johnson-medtech-announces-completion-of-first-cases-with-ottava-robotic-surgical-system
- Medtronic plc. Medtronic secures CE Mark for LigaSure RAS technology on the Hugo robotic-assisted surgery system. 15 Jul 2025. Available from: https://news.medtronic.com/Medtronic-shaping-future-of-surgery%2C-secures-CE-Mark-for-LigaSure-TM-technology-on-Hugo-TM-robotic-assisted-surgery-system
- Stryker. Stryker showcases next generation of Mako SmartRobotics at AAOS 2025 Annual Meeting. Apr 2025. Available from: https://www.prnewswire.com/news-releases/stryker-showcases-next-generation-of-mako-smartrobotics-at-aaos-2025-annual-meeting-302397425.html
- Zimmer Biomet. ROSA Knee System. Available from: https://www.zimmerbiomet.eu/en/products/rosa
- Globus Medical. ExcelsiusGPS Robotic Navigation Platform. Available from: https://www.globusmedical.com/international/solutions/excelsiusgps/
- Ruangsomboon P, Ruangsomboon O, Pornrattanamaneewong C, Narkbunnam R, Chareancholvanich K. Clinical and radiological outcomes of robotic-assisted versus conventional total knee arthroplasty: a systematic review and meta-analysis of randomized controlled trials. Acta Orthop. 2023;94:1–12.
- O'Neill N, Goh GS, Wong JYH, et al. Robotic-assisted total knee arthroplasty reduces radiographic outliers for low-volume, non-arthroplasty-trained surgeons. Arthroplasty Today. 2023;19:101167.
- Jayne DG, Pigazzi A, Marshall H, et al. Effect of robotic-assisted vs conventional laparoscopic surgery on risk of conversion to open laparotomy among patients undergoing resection for rectal cancer: the ROLARR randomized clinical trial. JAMA. 2017;318(16):1569–80.
- Ramirez PT, Frumovitz M, Pareja R, et al. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med. 2018;379(20):1895–904.
- Meara JG, Leather AJM, Hagander L, et al. Global surgery 2030: evidence and solutions for achieving health, welfare, and economic development. Lancet. 2015;386(9993):569–624.