Castro Laura1, Martin Janet2, Webb Steve3, Nepogodiev Dmitri1*, Glasbey James1*
*Joint senior authors
1Surgical Data Institute, Department of Applied Health Sciences, University of Birmingham, Birmingham, UK
2Department of Anesthesia and Perioperative Medicine, Schulich School of Medicine & Dentistry, University of Western, London, Ontario, Canada
3Australian and New Zealand Intensive Care Research Centre, School of Public Health and Preventive Medicine, Monash University, Melbourne, Australia
DOI: 10.5281/zenodo.20094836
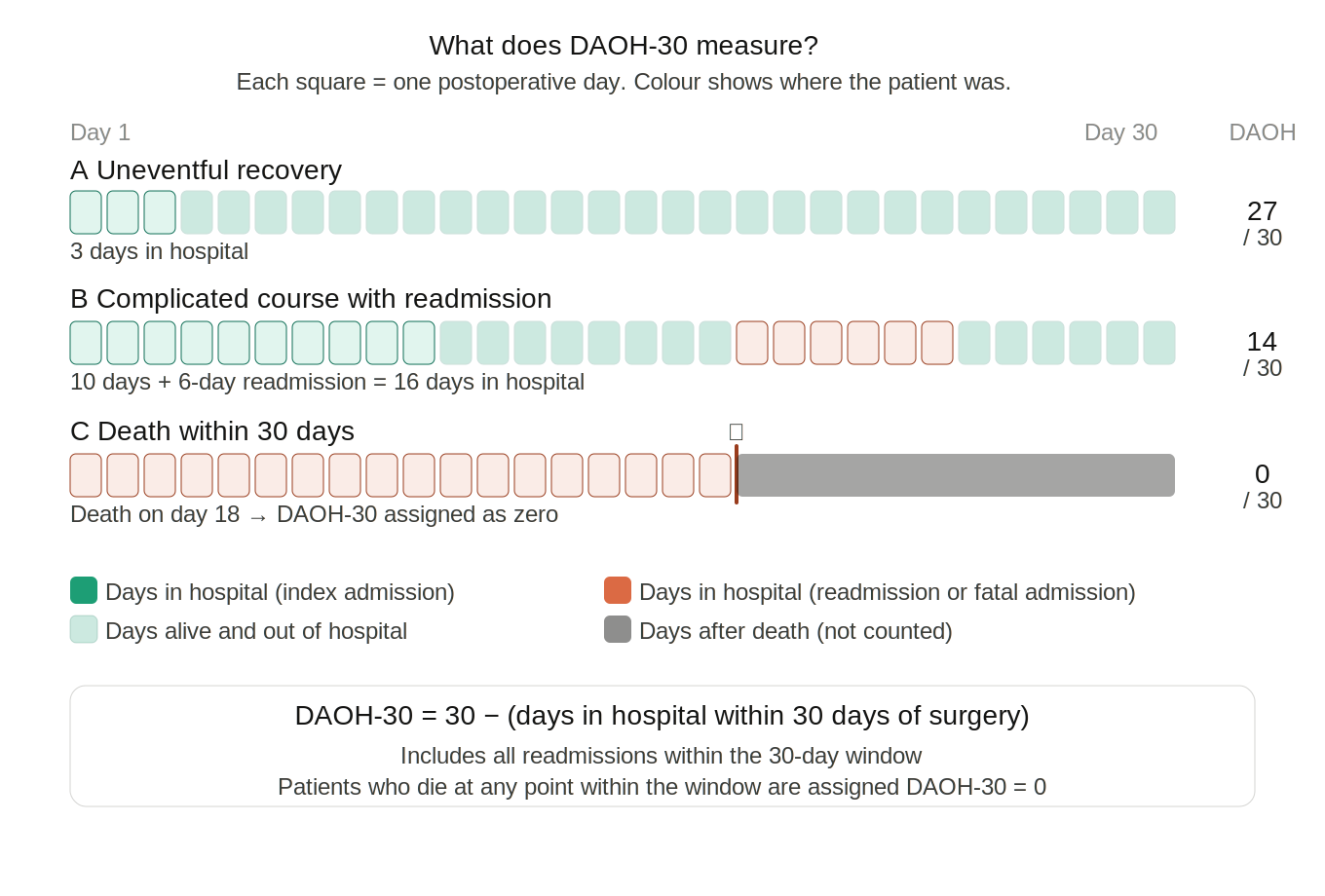
Over recent years, Days Alive and Out of Hospital (DAOH) has become established as a primary outcome in surgical and perioperative research, that serves as a proxy for patients’ return to function, survivorship, and more broadly for effective surgery and timely recovery. DAOH is the number of days, within a set postoperative period, that a patient has spent outside of the hospital1. It is therefore a composite of mortality and length of hospital stay (including readmissions). Patients who die within the set period are assigned ‘zero’ DAOH. Commonly, DOAH is reported within a 30-day (DAOH-30), 90-day (DAOH-90), or 1-year (DAOH-365) postoperative period. For example, DAOH-30 ranges from 0 to 30; a patient who has not been discharged following surgery by postoperative day 30 would be assigned 0, whereas a patient who had a day case operation with no readmission would be assigned 301.
An earlier iteration was Days Alive at Home (DAH), first validated in surgery by Myles et al.2 in 2017. Its validity was demonstrated in an Australian cohort of 2,100 surgical patients; both frailty and postoperative complications were associated with fewer days alive at home2. However, this metric had limitations, since discharge location is not always a patient’s home. In addition, it is challenging to collect DAH within electronic health records and administrative data, as discharge location is often not routinely recorded. DAOH addresses this limitation as it counts the number of days alive outside of the hospital, regardless of where the patient is discharged to. DAOH can be captured using simple routinely available data, such as the discharge date, making it an easy and efficient metric to capture, whilst still reflecting patients’ experience with healthcare resource use.
DAOH-30 has been now widely validated as a perioperative outcome in both elective and emergency surgery. Spurling et al.3, in a study of 79,000 patients who underwent emergency surgery, demonstrated that DAOH-30 is a sensitive marker for morbidity, with increasing risk correlated to fewer days at home (lower DAOH-30). In addition, DAOH-30 was strongly associated with increased one-year mortality. Jerath et al.1 reported similar findings in a cohort of 540,000 surgical patients: older, sicker patients consistently had lower DAOH, and those with complications experienced the lower DAOH.
Qualitative data also support the use of DAOH-30 as a patient-centred outcome. Researchers from Canada explored patients’ preference in treatment outcomes and found that the most important thing for patients after surgery was getting out of hospital, as this “signified normalcy, comfort and healing”, where recovery is accelerated4. Increased time spent at home has also been associated with better health-related quality of life and higher activities of daily living scores5. This highlights that DAOH can be a patient-centred outcome.
DAOH has been increasingly used in randomised controlled trials, with a recent systematic review identifying 19 trials with DAOH-30 as the primary outcome measure6. Critical and perioperative care trials using DAOH include the NOBLE7, HOT-ROX8 and VITAL9 trials. Furthermore, DAOH has been used to measure health inequalities; Boyle et al.10 identified that in New Zealand Māori patients who underwent a CABG operation experienced fewer DAOH than non-Māori patients.
There remain several limitations to DAOH-30. Firstly, it treats death and prolonged hospitalization as equivalent. A key area for development of DAOH is to explore how patients themselves weigh death against days spent in the hospital, and whether these weights differ by geography, demographics, or disease. Secondly, there are contextual challenges impacting the interpretation of DAOH-30. For example, DAOH-30 results may not be truly representative of health outcomes if patients have prolonged stays in hospital whilst ‘medically fit for discharge’ for non-medical reasons, such as waiting for social care services. Thirdly, DAOH-30 does not seek to measure the economic impact of surgery, which is a key consideration for patients and health systems11, 12. Consideration could be given to the integration of DAOH with utility scores, to enable its use in economic analysis.
An inherent limitation of DAOH is that it does not directly measure change in functional status and frailty once patients go home. Patients may experience serious decline in their function once they are discharged that prevents them from completing their previous care, work, and community roles. Some studies have sought to address this by collecting functional outcome data and weighting recovery trajectories, to better reflect post-discharge function and quality of life, particularly when DAOH is measured at longer timepoints13. This underscores the need for a suite of measures so that primary outcomes can be selected according to the specific research question and feasibility of data collection.
Looking forward, international collaborative efforts should be pursued to evolve DAOH-30 to improve validity and cross-contextual relevance to patients and communities.
Conflict of interest statement: All authors declare no conflict of interest.
Corresponding author: Laura, l.t.castro@bham.ac.uk, Surgical Data Institute, University of Birmingham, Birmingham B15 2TT, UK
References
- Jerath A, Austin PC, Wijeysundera DN. Days Alive and Out of Hospital: Validation of a Patient-centered Outcome for Perioperative Medicine. Anesthesiology. 2019;131(1):84-93.
- Myles PS, Shulman MA, Heritier S, Wallace S, McIlroy DR, McCluskey S, et al. Validation of days at home as an outcome measure after surgery: a prospective cohort study in Australia. BMJ Open. 2017;7(8):e015828.
- Spurling LJ, Moonesinghe SR, Oliver CM. Validation of the days alive and out of hospital outcome measure after emergency laparotomy: a retrospective cohort study. Br J Anaesth. 2022;128(3):449-56.
- Hladkowicz E, Auais M, Kidd G, McIsaac DI, Miller J. "I can't imagine having to do it on your own": a qualitative study on postoperative transitions in care from the perspectives of older adults with frailty. BMC Geriatr. 2023;23(1):848.
- Arya S, Langston AH, Chen R, Sasnal M, George EL, Kashikar A, et al. Perspectives on Home Time and Its Association With Quality of Life After Inpatient Surgery Among US Veterans. JAMA Netw Open. 2022;5(1):e2140196.
- Shackleton R, Vollam S, Gerry S, Shah A, Griffith DM. Reporting of Hospital-Free Days As an Outcome Measure in Critical Care Trials: A Systematic Review. Crit Care Med. 2025;53(12):e2686-e97.
- Birmingham Uo. Nitrous Oxide Management to Balance Healthcare and Environmental needs (NOBLE) Trial. IRAS ID 347203; 2024.
- Frei DR, Beasley R, Campbell D, Forbes A, Leslie K, Mackle D, et al. A vanguard randomised feasibility trial comparing three regimens of peri-operative oxygen therapy on recovery after major surgery. Anaesthesia. 2023;78(10):1272-84.
- Yeung J, Jhanji S, Braun J, Dunn J, Eggleston L, Frempong S, et al. Volatile vs Total intravenous Anaesthesia for major non-cardiac surgery: a pragmatic randomised triaL (VITAL). Trials. 2024;25(1):414.
- Boyle L, Curtis E, Paine SJ, Tamatea J, Lumley T, Merry AF. Using Days Alive and Out of Hospital to measure inequities and explore pathways through which inequities emerge after coronary artery bypass grafting in Aotearoa New Zealand: a secondary data analysis using a retrospective cohort. BMJ Open. 2025;15(2):e093479.
- Omar O, Kamarajah SK. Return to normal activity after abdominal surgery: a pre-planned secondary analysis of a randomised controlled trial across seven low- and middle-income countries. BMC Surg. 2025;25(1):399.
- Lawani I, Imorou Souaibou Y, Gnele JB, Lawani S, Bokossa C, Fadonougbo C, et al. Community consultation in a regional hospital in the Republic of Benin to co-develop the PUMA randomised trial: CEI in Benin. Impact Surgery. 2024;1(3):102-4.
- Delaney A, Tian DH, Higgins A, Presneill J, Peake S, Venkatesh B, et al. The Association Between Days Alive and Out of Hospital and Health-Related Quality of Life in Patients With Sepsis. CHEST Critical Care. 2023;Volume 1(Issue 3):100024.